|
12/13/2023 0 Comments Spike protein vaccine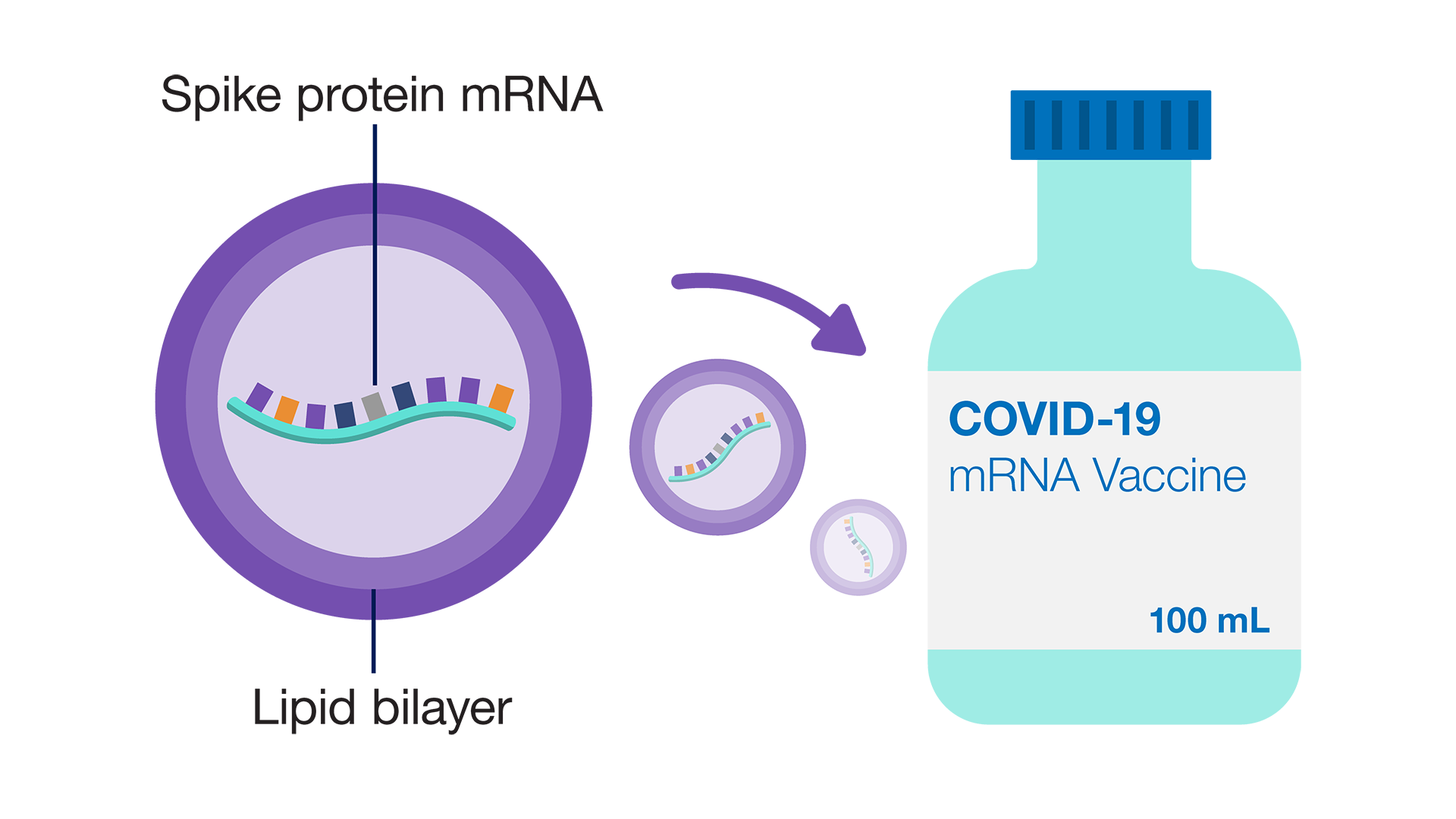
Two such traditional approaches relevant to COVID-19 vaccines-resulting in vaccines released for emergency use or in advanced clinical (phase III) trials-are whole inactivated virus (WIV) and protein-based vaccines targeting the spike protein or its receptor binding domain (RBD). 
While we anticipate that these important and exciting technologies will ultimately improve and become more adaptable to resource-poor settings in the future, for now we must continue expanding the production and distribution of vaccines that employ conventional technologies. This is a consequence of many factors, including difficulties in scale-up and scale-out technology transfer and production, costs, uncertain safety profiles, and intense antivaccine aggression on the internet ( 1, 2). Yet, the penetration of mRNA and adenovirus-vectored COVID-19 vaccines into low- and middle-income country (LMIC) markets is still modest. Their relatively quick turnaround, from the time the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) sequence appeared on a preprint server in January 2020 to production and clinical testing, could revolutionize vaccine development for future pandemic threats. Achieving these goals would also serve as an important reminder that we must continue to maintain expertise in producing multiple vaccine technologies, rather than relying on any individual platform.Ĭoronavirus disease 2019 (COVID-19) vaccines employing new technologies, including mRNA and adenovirus-vectored approaches, have generated intense public and scientific interest. There is an urgent need to continue accelerating these vaccines for LMICs in time to fully vaccinate these populations by the end of 2022 at the latest. Overall, these vaccines are exhibiting excellent safety profiles and in some instances have shown their potential to induce high levels of virus neutralizing antibodies and T cell responses (and protection) both in nonhuman primates and in early studies in humans. Several protein-based and protein particle-based vaccines are advancing with promising results. Three major WIV vaccines are beginning to be distributed widely. Vaccines based on whole inactivated virus (WIV) and protein-based platforms, as well as protein particle-based vaccines, are the most produced by LMIC vaccine manufacturing strategies. Immunizing the billions of people at risk for COVID-19 in the world's low- and middle-income countries (LMICs) still relies on the availability of vaccines produced and scaled through traditional technology approaches. 
The rapid development and deployment of mRNA and adenovirus-vectored vaccines against coronavirus disease 2019 (COVID-19) continue to astound the global scientific community, but these vaccine platforms and production approaches have still not achieved global COVID-19 vaccine equity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
